 Medical device manufacturers insist spinal-cord stimulators are safe - some 60,000 are implanted annually - and doctors who specialize in these surgeries say they have helped reduce pain for many of their patients. and as a treatment for an aging population in need of chronic pain relief. Companies and doctors aggressively push them as a safe antidote to the deadly opioid crisis in the U.S. 
“But look at me.”įor years, medical device companies and doctors have touted spinal-cord stimulators as a panacea for millions of patients suffering from a wide range of pain disorders, making them one of the fastest-growing products in the $400 billion medical device industry. “I thought I would have a wonderful life,” Taft said. Today, the 45-year-old Taft is virtually paralyzed, a prisoner in his own bed, barely able to get to the bathroom by himself. After an operation to repair it, he said, the device shocked him so many times that he couldn’t sleep and even fell down a flight of stairs. Taft’s stimulator failed soon after it was surgically implanted. It wouldn’t fix the nerve damage in his mangled right arm, Taft and his wife recalled the doctor saying, but a spinal-cord stimulator would cloak his pain, making him “good as new.” Federal law restricts this device to sale by or on the order of a physician.COLUMBIA, South Carolina (AP) - Desperate for relief after years of agony, Jim Taft listened intently as his pain management doctor described a medical device that could change his life. For complete indications for use, contraindications, warnings, precautions, and side effects, call 866.360.4747 or visit .Ĭaution: U.S. Your doctor may be able to provide additional information on the Boston Scientific Spinal Cord Stimulator systems. 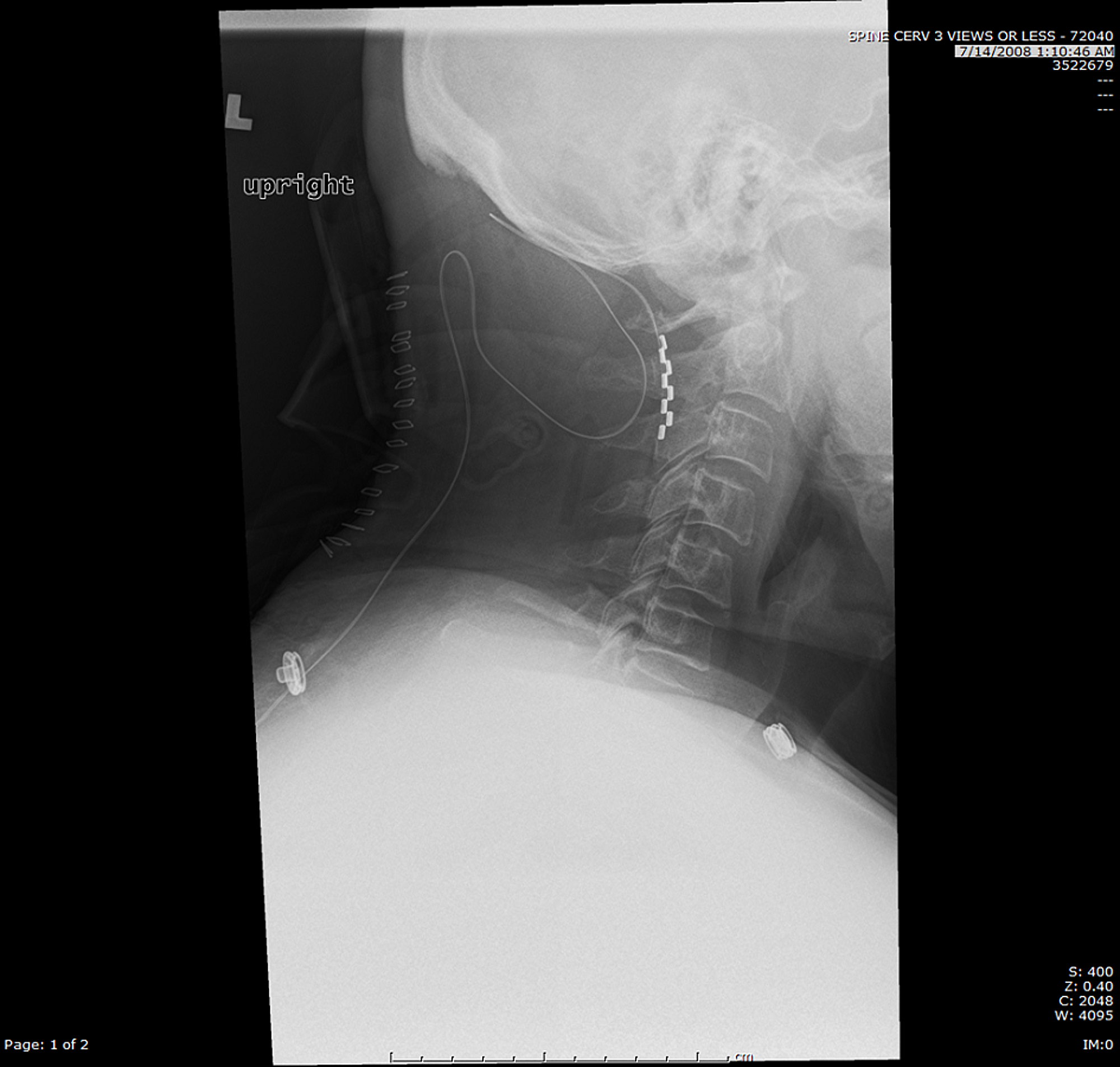
subperception therapy) it is less likely that sudden stimulation changes resulting in distraction could occur while having stimulation on when operating moving vehicles, machinery, and equipment. For therapy that does not generate paresthesia (i.e. Stimulation must be turned off first in such cases. Patients using therapy that generates paresthesia should not operate motorized vehicles such as automobiles or potentially dangerous machinery and equipment with the stimulation on. Advise your physician that you have a Spinal Cord Stimulator before going through with other implantable device therapies so that medical decisions can be made and appropriate safety measures taken. The Spinal Cord Stimulator system may interfere with the operation of implanted sensing stimulators such as pacemakers or implanted cardiac defibrillators. The system should not be charged while sleeping. Strong electromagnetic fields, such as power generators or theft detection systems, can potentially turn the stimulator off, or cause uncomfortable jolting stimulation. 
As a Spinal Cord Stimulation patient, you should not have diathermy as either a treatment for a medical condition or as part of a surgical procedure. Exposure to MRI may result in dislodgement of the stimulator or leads, heating of the stimulator, severe damage to the stimulator electronics and an uncomfortable or jolting sensation. Patients implanted with Boston Scientific Spinal Cord Stimulator systems without ImageReady™ MRI Technology should not be exposed to Magnetic Resonance Imaging (MRI). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
